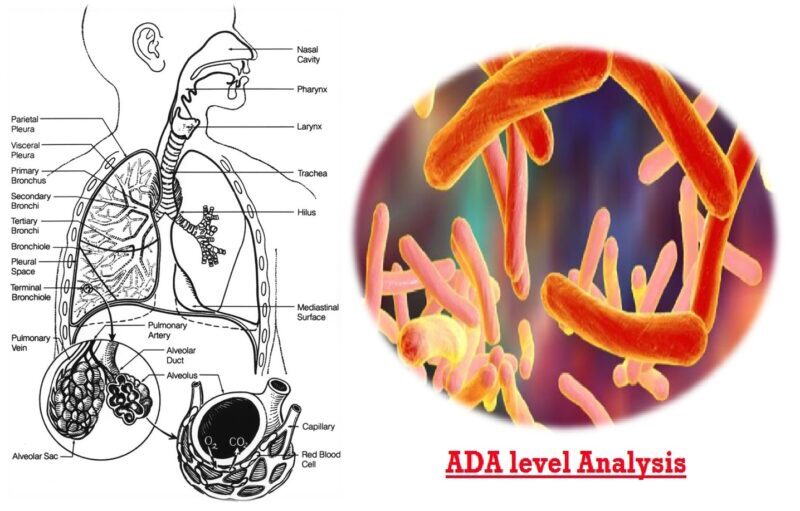
Adenosine Deaminase (ADA) is an endogenous tissue enzyme widely distributed in mammalian tissues, particularly in T lymphocytes. It is released into the serum of the patients with different types of malignancies and infections. It is associated with lymphocytic proliferations & differentiation and is a marker of cell mediate immunity. Two isoforms ADA1 & ADA2; ADA2 is the major contributor to the total ADA seen in TBM. Increased levels of ADA are found in various forms of tuberculosis, making it a marker for the same. The adenosine deaminase (ADA) test is not a diagnostic test, but it may be used along with other tests such as pleural fluid analysis, Acid-fast Bacillus(AFB) smear & culture, and/or tuberculosis molecular testing to help determine whether a person has a Mycobacterium tuberculosis infection of the lining of the lungs.
A culture is considered the "gold standard" for diagnosis of tuberculosis and guiding treatment, but it may take several days to weeks to complete. Molecular testing and the AFB smear are rapid tests, but they require that a sufficient number of bacteria be present in the fluid to detect them. Pleural fluid presents a unique problem with detecting M. tuberculosis because there may be a large volume of fluid with a very low number of bacteria present. Though the ADA test is not definitive, it is a rapid test and may be elevated even when there are few bacteria present. ADA results may be used to help guide treatment until results from a culture are available. The ADA test is used as an adjunct test to help rule in or rule out tuberculosis in pleural fluid. Though ADA is also increased in various infectious diseases like infectious mononucleosis, typhoid, viral hepatitis, initial stages of HIV, and in cases of malignant tumors, the same can be ruled out clinically.
Reference Values
-
- Serum, Plasma, Pleural fluid
- Normal : < 30 U/L
- Pericardial & Suspect : 30 U/L to 40 U/L
- Ascitic Fluids
- Strong Suspect : 40 U/L to 60 U/L
- Positive : > 60 U/L
- Normal : < 10 U/L
- CSF Positive : > 10 U/L
- Serum, Plasma, Pleural fluid
It is recommended that each laboratory establish its own normal range representing its patient population.
Principle
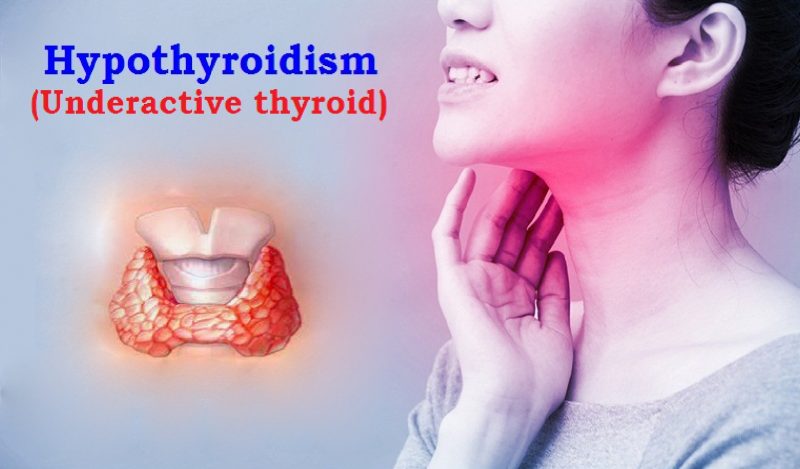
Adenosine deaminase hydrolyzes adenosine to ammonia and inosine. The ammonia formed further reacts with a phenol and hypochlorite in an alkaline medium to form a blue indophenol complex with sodium nitroprusside acting as a catalyst.The intensity of the blue colored indophenol complex formed is directly proportional to the concentration of ADA present in the specimen.
Requirements
- ADA-MTB Reagent comprises of:
- ADA-MTB reagent (L1)—Buffer reagent.
- ADA-MTB reagent (L2)—Adenosine reagent.
- ADA-MTB reagent (L3)—Phenol reagent.
- ADA-MTB reagent (L4)—Hypochlorite reagent.
- ADA-MTB standard (S)—ADA standard.
Store the kit at 2–8oC, away from light. Stability of the kit is as per the expiry date mentioned on the label. It is important that kit components from the same lot are used for achieving accurate and reproducible results. Do not intermix reagents from different lots.
Reagent Preparation :
-
- Reagents L1, L2 and standard are ready to use.
- Adenosine. Reagent (L2) may form crystals at 2–8°C. Dissolve the same by gently warming (37 to 50°C) the Reagent for some time before use.
- Both the Phenol Reagent (L3) and Hypochlorite Reagent (L4) need to be diluted 1:5 with Distilled water before use (1 part of Reagent + 4 parts of distilled water). The Working Phenol Reagent and Working Hypochlorite Reagent are stable for at least 6 months when stored at 2–8°C in tightly closed bottles.
- The sequence of addition of Reagents should be followed meticulously for achieving accurate results.
- Test tubes with test tube stand.
- Water bath/Incubator (37°C).
- Distilled or deionized water.
- Variable volume pipettes.
- Spectrophotometer with filter at 570–630 nm (Hg 578 or 623 nm) at 37°C or colorimeter with yellow or red filter.
- Stopwatch.
Specimen Collection & Preparation
Collect the specimen prior to use of antimicrobial agent. Wherever possible, indicate clearly that patient is on antitubercular drugs.
-
- Cerebrospinal Fluid : Collect as much as possible in a syringe, clean skin with alcohol before aspirating specimen.
- Body fluids : Disinfect the site and collect specimen with aseptic precautions.
- Serum, Plasma : No special preparation of the patient is required prior to sample collection by approved techniques. It is recommended to use fresh sample specimen for testing. Do not use hemolyzed, contaminated or turbid sample specimens. Fresh EDTA, citrate, heparinized or oxalate anticoagulated (dry anticoagulant) plasma specimens are suitable for performing the test. ADA is reported to be stable in serum for 3 days at 2–8°C and in biological fluids for 2 days at 2–8°C, as after this, ammonia may be released in the samples even without any microbial contamination.
Test Procedure
- Bring all Reagents and samples to room temperature before use.
- Prepare the working phenol reagent and working hypochlorite reagent.
- Set the spectrophotometer filter at 570–630 nm (Hg 578 or 623 nm) at 37°C.
- Pipette into clean dry test tubes labeled Blank (B), Standard (S), Sample Blank (SB) and Test (T) as follows:
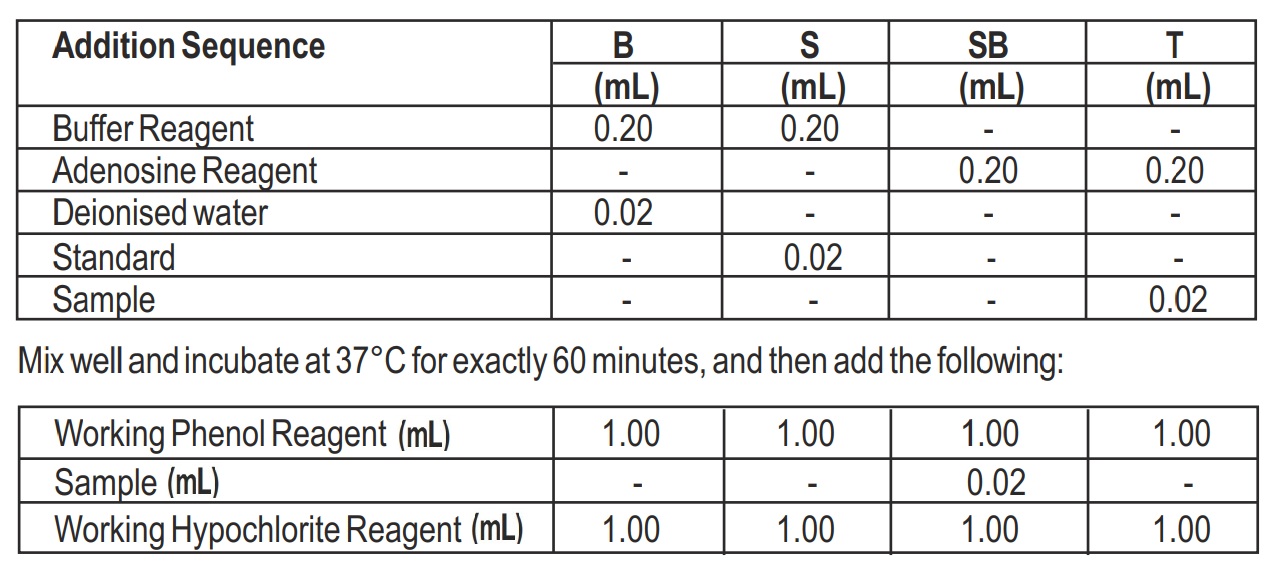
- Mixed well and incubate at 37°C for 15 minutes or at Room temperature for 30 minutes.
- Measure the absorbance of the Blank (B), Standard (S), Sample Blank (SB) and Test (T) against Distilled water and calculate as follows..
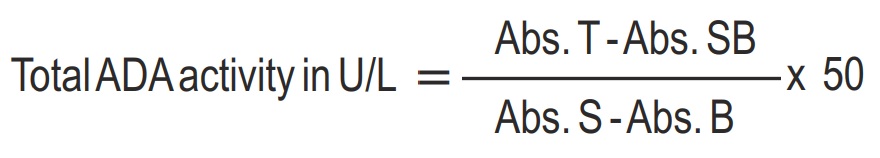
- The procedure is linear up to 150 U/L. If values exceed this limit dilute the sample with deionized water and repeat the assay. Calculate the value using the appropriate dilution factor.
Special Note :
-
- One unit of ADA activity releases three nanomoles of ammonia in the reaction in 1 hour at 37°C.
- Patients with hyperammonemia, kidney disorders and hepatitis can present high level of ADA values.
- Patients with chronic malnutrition or HIV can present low levels of ADA values.
- Higher levels of ADA are also found in leprosy, brucellosis, HIV infections, viral hepatitis, infectious mononucleosis and liver cirrhosis. Before arriving to a diagnostic decision, these clinical conditions must be ruled out.
- Using a cut off level of 60 units/L of ADA, values has been reported to show the specificity and the sensitivity of the test as above 90% for the MTB infection.
- Below 60 U/L of ADA, the serum of ADA specificity and sensitivity is lower and should be interpreted in the light of other tests for confirmation of Mycobacterium tuberculosis infection.
Conscious about Interpretation of Result
For False positive results :
-
- False Positive result may occur in case of addition of too much sample in relation to reagent. For this case add exact quantity of sample as mentioned in the package insert.
- Markedly lipemic and contaminated sample could give improper reading, hence don't use lipemic and contaminated sample for testing it's can give False Positive result.
- Less amount of standard added in the respective tube can give False Positive result. So added exact quantity of standard as mentioned in the test literature.
- Incubation period or incubation time must be maintained, exceeded incubation give False Positive result. Hence, ensure 1st incubation exactly at 37°C and 2nd incubation at 37°C or at RT for exact period not exceeded as mentioned in the test kit.
- Correct interpretation of results is very important, So, read the result within 30 minutes after performing the test as per instructions given in the literature.
For False negative results
-
- Addition of less amount of sample with respect to the reagent can give false negative results. So add exact quantity of sample as mentioned in the kit for true value.
- Addition of higher amount of standard in the respective tube can produce false negative results; hence add exact quantity of standard as mentioned in the test kit for evolution of true result.
- Incubation period or incubation time must be maintained, Improper incubation give False Negative result. Hence, ensure 1st incubation exactly at 37°C and 2nd incubation at 37°C or at RT for exact period as mentioned in the test kit.
- Improper dissolution of the adenosine reagent can give false negative results, So dissolve the adenosine reagent properly by gently warming at 37–50°C in water bath for the evolution of true ADA level.
- Improper mixing of the sample with reagent may give false negative results Mix the sample properly with reagent after every addition.
For Reagent not working
-
- Improperly mixed reagent can give false positive results or false negative results. So bring the reagent at RT prior to the test. Mix the reagent properly by tilting the vial upside down before performing the test.
- Improper dissolution of the adenosine reagent can give false positive results or false negative results, So dissolve the adenosine reagent properly by gently warming at 37–50°C in water bath for the evolution of true ADA level.